Help Aids Top
Application:
This procedure computes sample size for
non-inferiority and superiority tests in 2×2 cross-over designs in which the
outcome is a continuous normal random variable. A non-inferiority test tests that
the treatment mean is not worse than the reference mean by more than a small
equivalence margin. The actual direction of the hypothesis depends on the
response variable being studied. A superiority test tests that the treatment
mean is better than reference mean by more than a small equivalence margin. The
actual direction of the hypothesis depends on the response variable being
studied. The following hypotheses are usually considered: ![]() .
.
Procedure:
- Enter
a) value of α, the probability of type I error
b) value of β, the probability of type II error
c)
value of allowable difference
d) value of population variance
e) value of δ>0, the superiority margin or value of δ<0, the non-inferiority margin.
- Click the button “Calculate” to obtain result sample size of each group n.
Formula:
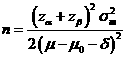 (*)
(*)
Notations:
α: The
probability of type I error (significance level) is the probability of rejecting the true null
hypothesis.
β: The
probability of type II error (1 – power of the test) is the probability of
failing to reject the false null hypothesis.
δ: Clinically meaningful difference. The largest change
from the reference value (baseline) that is considered to be trivial.
μ2 – μ1:
The true difference between the two mean values at which the power is calculated.
Population variance:
The within mean square error from
the ANOVA table used to analyze the Cross-Over design.
Examples
Example 1: Suppose you want
to consider the sample size of a balanced, cross-over design that will be
analyzed the t-test approach. Thus when the non-inferiority margin is δ=-0.05, the true difference between the
means under H0 is assumed
to be 0, with population variance ![]() of 0.01 and the
significance level of α=0.05 given.
We have the sample size to achieve 80% power for both values of at β=0.2 is n=13.
of 0.01 and the
significance level of α=0.05 given.
We have the sample size to achieve 80% power for both values of at β=0.2 is n=13.
Reference: Chow, Shao and Wang, Sample Size Calculations In Clinical
Research,