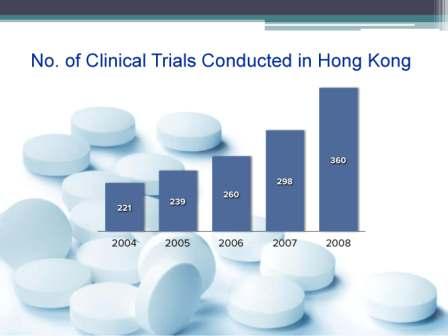
The Centre for Clinical Research and Biostatistics – Clinical Trials Registry (CCRBCTR), provides all prospective registrants with a convenient means to submit trial registration for all countries. Registered information is displayed in English only (except for the Chinese study title) and is accessible to the public at no charge. Fees would only apply to services other than clinical trials registration, such as translation of registered information. All trial registrations are electronically searchable with an in-house mechanism to ensure the validity of the registration data. With the updated webpage, the registrants can manage all their registrations with one login account and the public can access the audit trail for all registration amendments.
Since December 2009, the Centre for Clinical Research & Biostatistics, Clinical Trials Registry – CUHK is a Partner Registry of a WHO Primary Registry – Chinese Clinical Trials Registry (ChiCTR).We gather trial information and make the information available in the WHO trial registration dataset, so trials registered on our registry are now also registered on ChiCTR with registration numbers from the WHO Primary Registry and all registered information in CCRBCTR is publicly searchable at WHO ICTRP and ChiCTR, Centre for Clinical Research and Biostatistics, Clinical Trials Registry of Chinese University of Hong Kong is now in compliance with WHO ICTRP and ICMJE trial registration requirements and it satisfies the requirements of many major journals.
Clinical Trials Registration Instruction
User Account Registration
Clinical Trial Registry User Login
View Clinical Trial Registration