Help Aids Top
Application: The
determination of the number of patients needed in a prospective comparison of
survival curves, when the control group patients have already been followed for
some period, following hypotheses are usually used: ![]()
Procedure:
- Enter
a) value of α, the probability of type I error
b) value of β, the probability of type II error, or (1-power) of the test
c) value of δ, the minimum hazards ratio, it is specified in alternate hypothesis
d) value of MS, the control median survival (year), it is estimated
from existing control data
e) value of QC, the proportion of patient in control group compared with
experimental group
f) value of QE, the proportion of patient in experimental group compared with control group, where QC + QE = 1
g) value of T0, accrual duration (year), the length of time to recruit patients for
study.
h) value of T-T0, follow-up duration (year), the length of study time of all recruited patients to the end of study T
- Click the button “Calculate” to obtain the sample size of patients in the experimental arm and the total number of deaths at the end of study
Assumption:
1.
Time
to survival is exponential distributed with hazard rate λ.
Formula:
Consider
that there are two independent groups of sizes nC
and nE
all followed to the terminal event where time t is measured from the time of entry into the study.
Under
null hypothesis,![]() , test statistic is
, test statistic is![]() , where
, where![]() and
and![]() are the estimated hazard rates,
are the estimated hazard rates, 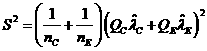 , and
, and![]()
For the
determination of sample size and power one specified the minimal relevant difference![]() , which yield
, which yield![]() . Under the null hypothesis,
. Under the null hypothesis,![]() and
and ![]() where
where![]() .
. ![]() is defined as
is defined as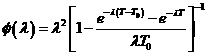
The
sample size to achieve specified significance level and power is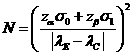 , where
, where![]() is the upper 100(1-p) percentile
of the standard normal distribution.
is the upper 100(1-p) percentile
of the standard normal distribution.
Here the calculator uses two-sided test. α of the above equation is replaced by α/2.
Notations: α: The
probability of type I error (significance level) is the probability of rejecting the true null
hypothesis. β: The
probability of type II error (1 – power of the test) is the probability of
not rejecting the false null hypothesis. Example: Consider that a clinical trial is to be
conducted for a disease with moderate levels of mortality with hazard rare
λ=0.30, yielding 50% survivors after 2.3 years (MS=2.3). Suppose that with treatment we are interested in a reduction in
hazard to λ=0.20, i.e., an increase
in survival to 64% at 2.3 years (δ=0.3/0.2=1.5). With equal-sized group (QC=QE=0.5), significance level
test (α =0.05) with power 90% power (β=0.1), and assume that
recruitment was to be terminated after 3 years of a 5-year study (T=5, T0=3,
T-T0=2), then the required
sample size is approximate 461 (N=461)
and the total number of deaths is 264 (ND=461). Reference: Lachin (1981) Controlled Clinical Trials 2: 93-113